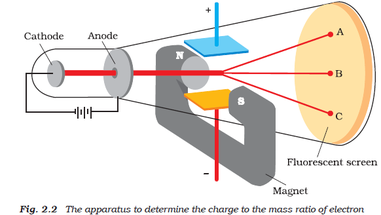
In 1909, more information about the electron was uncovered by American physicist Robert A. Thomson was an English physicist who worked with cathode ray tubes similar to those used by Crookes and others in the mid-19th century. Millikan's Oil-Drop Experiment: Electron Charge (credit a: modification of work by Nobel Foundation credit b: modification of work by Eugen Nesper credit c: modification of work by “Kurzon”/Wikimedia Commons). Simultaneous deflections by applied electric and magnetic fields permitted Thomson to calculate the mass-to-charge ratio of the particles composing the cathode ray. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Thomson produced a visible beam in a cathode ray tube. Everett who helped to greatly increase Thomson's experimental range.\) (a) J. 
About 1894 he acquired an excellent glassblower named E. Thomson‘s historic Royal Institution lecture of April 30, 1897, on Cathode Rays, has been reproduced in full in the November-December issue of the American Journal of Physics as the contribution of the American Association of Physics Teachers to the Electron Jubilee celebrations (see also Nature, 160, 776 1947). He was very fumble fingered and had a tendancy to break things. Incidently, Thomson was a very unhandy person. He placed cathode tubes in electric and magnetic fields. I am grateful to my supervisor,David Gooding for encouragement and helpful criticism and to the University of Bath for financial support. Thomson was a physics professor at Cambridge University in the UK. Thomsons work on gaseous discharge,’ University of Bath, 1985, in which further details may be found. 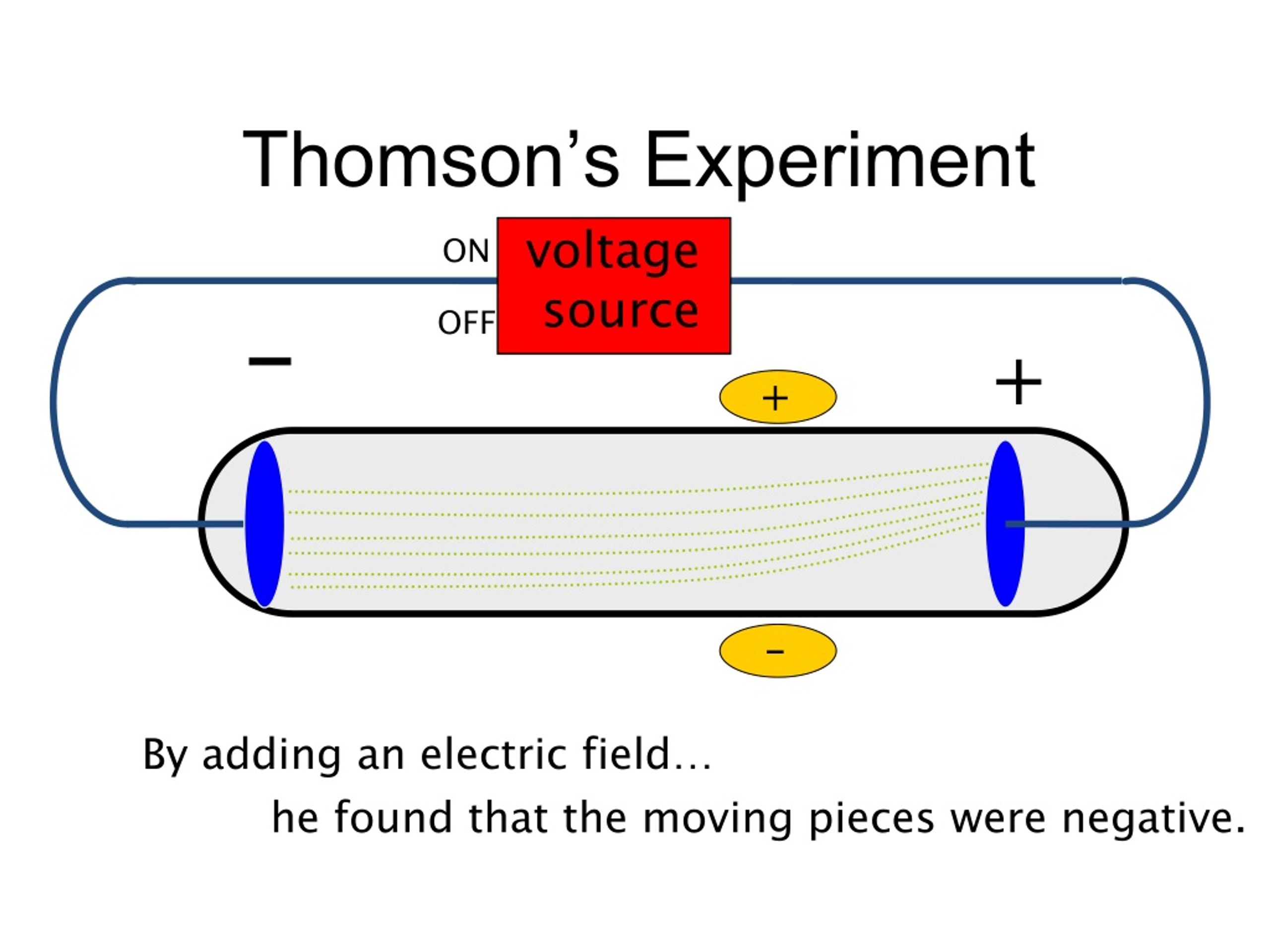
3: The cathode ray tube was first invented. This paper is condensed from my Ph.D thesis ‘Theory and experiment in J.J. Crookes concluded that the cathode ray was made of particles which must have mass. Thomson was the first to recognize that all the cathode rays have the same. Notice that the cathode and anode are positioned so that the rays will strike the top of the paddle wheel. 17), British physicist, president of the Royal Society, teacher of Ernest Rutherford and winner of the Nobel Prize in physics in the year 1906, succeeded in deciphering the nature of the cathode rays by conducting different experiments. The amount the cathode ray bent from the straight line using either the electric field or the magnetic field allowed Thomson to calculate the e/m ratio. Three experiments led him to this.: irst, in a variation of an 1895 experiment by Jean Perrin, Thomson built a cathode ray tube ending in a pair of metal cylinders with a slit in them. Upon starting up the cathode ray tube, the wheel rotated from the cathode towards the anode. (d) An isotope of iodine is used for making tincture iodine, which is used as a medicine. This allowed him to use either electrical or magnetic or a combination of both to cause the cathode ray to bend. In the -scattering experiment, a gold foil. Thomson also could use magnets, which were placed on either side of the straight portion of the tube just to the right of the electrical plates. The two plates about midway in the CRT were connected to a powerful electric battery thereby creating a strong electrical field through which the cathode rays passed. The long glass finger (in the photo) projecting downward from the right-hand globe is where the entire tube was evacuated down to as good as a vacuum as could be produced, then sealed. Thomson in 1897 announcing the discovery of the electron. Thomson was awarded the Nobel prize in 1906 for discovering the fundamental particle electron. The mass of the electron is 9.1094 x 10 - ³¹ kg. With Thomson’s mass to charge ratio and charge on a single electron, Millikan calculated the mass of an electron. As regards the radiant efficiency, that was found to be. According to him, the charge of a single electron was 1.6 x 10 -19 Coulomb. Thomson’s experiments was clearly stated in the introduction to his 1897 paper. His 1897 experiment on cathode rays is generally regarded as the discovery of the electron. Th diagram below appeared in an article by J.J. cathode rays and the particles discharged from negatively - charged bodies. In this section I will discuss the grounds for belief in the existence of the electron by examining J.J. It is about one meter in length and was made entirely by hand. The image below of a CRT used by Thomson in his experiments. Only the end of the CRT can be seen to the right-hand side of the picture. Using more than one cathode-ray tube in 1897 for his experiments, Thomson managed to identify a particle 1,000 times smaller than the then known smallest piece of matter: a hydrogen atom. Thomson and a cathode ray tube from around 1897, the year he announced the discovery of the electron. Thomson used results from cathode ray tube (commonly abbreviated CRT) experiments to discover the electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
